What is: Enzymatic Browning?
|
Have you ever wondered why those bananas you bought last week when they were perfectly yellow have started to develop brown spots? Or perhaps why that apple you cut into slices hours ago and neglected has started to take on an unappetizing brown color? The answer, of course, is enzymatic browning. This chemical process is due to enzymes in our food which, when exposed to oxygen, create color pigments and a myriad of flavor components, both desirable and... perhaps not-so-desirable.
Understanding the process is the first step in using it for the good of food, instead of for its detriment! |
The Chemistry
What is happening here? Now, to understand the chemistry of this process, the players at work must first be defined. Here is a list of the players involved; keep these in mind when reading through the next section:
- Polyphenols
- Polyphenol Oxidase (PPO)
- Catechol Oxidase
- Melanin
- Oxidation Products
What is Involved?
|
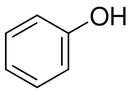
The molecule on the left is the general structure of a phenol. While they exist in food as simplistic forms such as this, they are generally involved in much more complex structures known as polyphenols. As the name suggests, they are large chemical structures that include multiple copies of the phenol ring on the left.
These polyphenol structures often act as anti-oxidants in plants, and often are responsible for their pigmentation. |
What is Going On?
|
Polyphenol oxidase, commonly abbreviated as PPO, is a class of naturally occurring enzymes that occur commonly in plants and animals. Their role in natural systems is typically as a form of resistance to microbial infection, and their effect on our food is more of a secondary one. The figure on the right shows the two possible chemical reactions that this particular enzyme catalyzes in the presence of oxygen. Starting from the naturally occurring polyphenols mentioned above, the aromatic rings become progressively oxidized, resulting in melanin, as well as other flavor compounds. Melanin is a product that gives this reaction its characteristic brown color. |
|
Other enzymes, such as catechol oxidase and tyrosinase, also contribute to this reaction. The exact chemical mechanisms vary based on what food is undergoing the reaction, but the results are typically the same: a darkening of color and the generation of new chemical compounds.
I'm sure you've all seen something like this before!
Good or Bad?
Now, in most cases, those brown spots on your banana mean you should think about eating them soon. Enzymatic browning is often detrimental to a large portion of fruits, vegetables, and seafood. But don't be too quick to judge! This process is also known to produce a variety of important flavor components that give tea its distinct flavor and aroma. While many of the flavor compounds are created during the Maillard Reaction, enzymatic browning also plays an important role in the production of tea. In some darker teas, such as black tea, the leaves are crushed up upon picking, thereby increasing the rate of oxidation and the rate of color/flavor formation.
So the next time you take sit down to relax and sip a cup of tea, remember to be thankful for those wonderful little enzymes!
So the next time you take sit down to relax and sip a cup of tea, remember to be thankful for those wonderful little enzymes!
Steps to Avoid Browning

There are a variety of ways to prevent those freshly cut apples from turning brown, and many of them are simple and easy to do at home. The list below are just some of the more common methods used, but anything that can prevent the reaction above from taking place will work. If you knock out any of the required components (the enzyme, substrate, or oxygen), then the reaction will not occur.
A few common methods:
A few common methods:
- Wrapping up your apple slices, or equivalent fruit, can help prevent the oxygen in the surrounding air from coming in contact with the newly exposed surface. By keeping oxygen away from the enzymes responsible for the oxidation of polyphenols, the mechanism of browning can be slowed. This can be achieved easily at home through the use of plastic (saran) wrap, or a Ziploc bags.
- Citric acid can also be used to prevent browning by denaturing enzymes such as polyphenol oxidase, thereby halting the process. This can be achieved easily by squirting a small amount of lemon juice on exposed surfacess.
- Blanching is another method to prevent oxidation. For those unfamiliar with the process, blanching involves quickly submerging the the fruit of choice into boiling water for a short amount of time. This is followed by a quick plunge into ice cold water. Similar to how citric acid denatures enzymes by lowering the pH, increased temperature also works to unravel the proteins that seek to oxidize our food.
- Salt water is also an option, though too much can adversely affect the taste of the fruit. A light amount of salt can help kill any bacteria or fungi that would take host on the surface, slowing degradation.
References
- "Enzymatic Browning." Food-Info.13 May 2013. Web. 1 Mar. 2014. <http://www.food-info.net/uk/colour/enzymaticbrowning.htm>
- How to Blanch: Blanching Fruits and Vegetables. About, Web. 11 Mar. 2014. <http://localfoods.about.com/od/preparationtips/qt/How-To-Blanch.htm>.
- Lee, Chang Y., and John R. Whitaker. Enzymatic Browning and Its Prevention. Vol. 600. American Chemical Society, 1995
- Nokthai, Prontipa, Vannajan Sanghiran Lee, and Lalida Shank. Molecular Modeling of Peroxidase and Polyphenol Oxidase: Substrate Specificity and Active Site Comparison. International Journal of Molecular Sciences. 1 Mar. 2014. <http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2956093/>
- "Polyphenol Oxidase." Worthington Biomedical Corporation. Web. 1 Mar. 2014. <http://www.worthington-biochem.com/ty/default.html>