What is a: Leavener?
While matzo and tortillas certainly have their place in the culinary playground, most baked goods are expected to rise to the occasion. The mechanism that causes a relatively flat and uninteresting piece of dough to expand and grow into an elegant loaf of bread is one of simple chemistry.
Culprit 1: Yeast
|
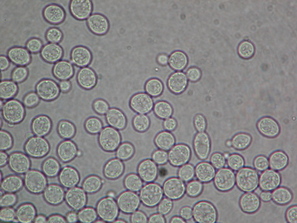
When people hear the term "yeast", they typically think of one species of funny little organisms that somehow help make bread rise. The term yeast is somewhat ambiguous, as it actually refers to a collection of around 1,500 species of microorganisms that are classified under the Fungi kingdom. The image on the right is of Saccharomyces cerevisiae, the species of yeast most commonly used for baking, beer brewing, and wine making.
If you've ever baked something from scratch, chances are you've heard of these little guys! Now the question stands: how do these mischievous little critters make flatbread rise up in defiance of gravity and stand tall! |
What is Going On?
|
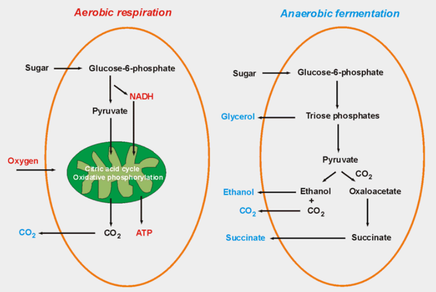
Yeast can undergo two different metabolic pathways dependent on the type of environment they are placed in. Aerobic respiration occurs when there is oxygen present, and anaerobic fermentation occurs when oxygen is not present. The general reactants and products are outlined by the image on the left (click to magnify). The main product that we are interested in is carbon dioxide, which leads to the formation of air bubbles in our dough. These air bubbles are trapped by the protein backbone formed by the other ingredients, leading to a rise. Over time, this leads to a tall and proud loaf of bread instead of a flat one! |
The aerobic pathway is the one typically used for baking, but the anaerobic pathway also has its place. Fermentation is necessary for the formation of ethanol, without which our beers and wines would never be the same! So be thankful for these little, mysterious creatures because they are essentially the unsung heroes of some of the most popular baked goods and alcoholic beverages.
Types of Yeast
While there exist many different types of yeast that can be utilized by beer makers, wine distillers, and scientists alike, bakers place the yeast they use into 3 general categories: Active Dry Yeast, Instant Yeast, and Fresh Yeast. They all have their own unique applications, and understanding when to use them is important in making the most of these microorganisms. All types of yeast require storage in a cool, dry place.
Active Dry Yeast is the type most often used by bakers. This type of yeast is sold in a dormant state, from which it must be awoken if it is to be used. This requires what is known as "proofing", which involves putting the yeast into a warm water bath with sugar. If done properly, these guys will be up and bubbling in no time!
Instant Yeast is the second type of yeast used, and is unique in that it does not require proofing. This type of yeast is typically more concentrated than active dry yeast, and is used in similar applications as the first type.
Fresh Yeast is sold wet, instead of dry. It is much harder to work with than other forms of yeast, and is typically used by professional bakers. Similar to active dry yeast, it needs to be proofed before being put to use.
Active Dry Yeast is the type most often used by bakers. This type of yeast is sold in a dormant state, from which it must be awoken if it is to be used. This requires what is known as "proofing", which involves putting the yeast into a warm water bath with sugar. If done properly, these guys will be up and bubbling in no time!
Instant Yeast is the second type of yeast used, and is unique in that it does not require proofing. This type of yeast is typically more concentrated than active dry yeast, and is used in similar applications as the first type.
Fresh Yeast is sold wet, instead of dry. It is much harder to work with than other forms of yeast, and is typically used by professional bakers. Similar to active dry yeast, it needs to be proofed before being put to use.
Culprit 2: Chemical Leaveners
There are some bakers who prefer to use chemical leavening agents. This works on a similar principle seen in the naked egg experiment, so check out that page for a hands on demonstration. Basically, the reaction occurs between an acid and a base to produce carbon dioxide in a similar fashion to how yeast work. There are a few different types, but they all operate in the same way.
|
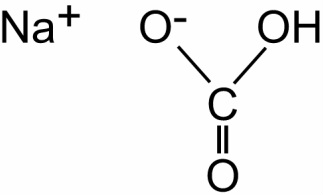
Baking Soda is perhaps the most well known. This type of leavener is typically only used when there is already an acid present in the recipe. Baking soda is also known by the name sodium bicarbonate, the chemical structure of which can be found on the right. This is common in grocery stores, and most of you probably already have this in your kitchen for a myriad of other applications.
|
Baking Powder is another common leavening agent. In the same manner as baking soda, this leavener works through an acid-base reaction. The difference between the two is that baking powder comes with acid build in! As soon as moisture is added (in the form of water, milk, etc.), the reaction begins and carbon dioxide is produced. There are multitude of different acids and bases that can be used in what is known as "baking powder", but they all function under the same principles of chemistry to breath life into our baked goods!
The Conclusion
Whether you choose to use yeast, or a chemical leavener, hopefully you will remember the main reason that your dough is rising: carbon dioxide! The way that this gas is created may change with your choice, but the product is the same. If it weren't for these various methods, our breads would be flat and our beer/wine tasteless, so be thankful for those who learned to harness the power of chemistry of the good of food lovers around the world!
References
- Feldmann, Horst. Yeast Metabolism. Adolf-Butenandt-Institute, Web. 10 Mar. 2014. <http://biochemie.web.med.uni-muenchen.de/Yeast_Biol/03%20Yeast%20Metabolism.pdf>.
- Phillips, Sarah. Leaveners. Sarah Phillips, 2000. Web. 15 Mar. 2014. <http://baking911.com/learn/ingredients/leaveners>.
- Stewart, Kelly. The science of baking: Kitchen chemistry. Culinate, 2009. Web. 7 Mar. 2014. <http://www.culinate.com/articles/features/baking_chemistry>.
- The world of leaveners. King Arthur Flour, Web. 5 Mar. 2014. <http://www.kingarthurflour.com/recipe/leavens.html>.
- Understanding Baking: How Yeast Works. The Prepared Pantry, Web. 7 Mar. 2014. <http://www.preparedpantry.com/howyeastworks.aspx>.
- Yeast. HowStuffWorks, Web. 14 Mar. 2014. <http://science.howstuffworks.com/life/fungi/yeast-info.htm>.