What is: The Maillard Reaction?The maillard reaction is chemical reaction that occurs on the surface of our foods during cooking, resulting in the myriad of flavors, colors, and aromas that result. It is a complex process, but one that is definitely worth knowing!
This reaction was discovered by Louis Camille Maillard in the early 1900's, and has been instrumental in understanding the hundreds of different flavor and aroma compounds that are created during the cooking process. |
The Chemistry
|
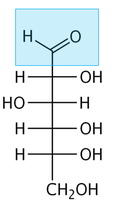
Sugars are the first component in this reaction (reducing sugars to be specific). The structure on the left is glucose, a common sugar. The carbonyl group of the sugar is highlighted in blue, and is the portion of the molecule that reacts.
As you may already know, sugars are common in our food and in our bodies. They exist in many forms (not just the sugar on the grocery store shelf!). In the context of this reaction, the necessary chemical structure is that of a reducing sugar. What this means is that the sugar must contain an aldehyde (which is illustrated by the blue highlight on the left). This portion of the molecule is reactive, and will undergo nucleophilic attack from the next component of the reaction . |
|
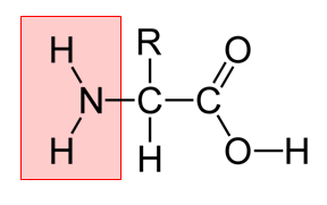
Amino acids are the next component in this reaction. They are buildings blocks for proteins which exist in our bodies, and in our food. The amino group (highlighted in red) is what acts as a nucleophile, attacking the carbonyl group of the reducing sugar. This results in a new compound. From here, the resulting compound undergoes a reaction known as the amadori rearrangement, followed by a a myriad of different reactions including dehydration, fission, and Strecker degradation. While the specifics behind these reactions are not well characterized by scientists, the resulting compounds are what give our food their flavor and color!
|
What is Involved?
- Time plays a big role in this reaction, as you may already expect. The longer you are heating your food, the further the maillard reaction will progress. Just make sure not to cook you food too long, or you may end up burning it!
- Temperature is another decisive factor in this reaction. The maillard reaction can technically occur at any temperature, but it is only noticable when accelerated by heat.
- Moisture is a component of the reaction that may be less obvious. This reaction will not occur under an environment with a high water content. This means that boiling those carrots is not causing them to undergo the maillard reaction, so none of the tasty flavor products are produced!
- pH is another important factor. This reaction occurs fastest under a basic environment favoring nucleophilic attack. Pretzels are an interesting example that you may be familiar with. Pretzels are typically in lye, which raises the pH to a great extent and gives them their dark brown exterior.
The Results
|
The products of this complex set of reactions are easy to see. For one, melanoidin is produced, which gives our food its distinct brown color. Those sear lines on your steak? Those are caused by these newly formed color pigments. There are hundreds of different flavor compounds that are created during this reaction, giving our foods their own unique flavor profile. The aromatic composition of our food is also defined by this process. For example, 6-acetyl-2,3,4,5-tetrahydropyridine is responsible for the smell of baked goods. If you've ever walked into a bakery, you'll know this smell! The next time you are cooking in the kitchen, remember that the maillard reaction is most likely responsible for your delicious food! |
References
- Acrylamide in Food and Cancer Risk. National Cancer Institute. Web. 31 Jan. 2014. <http://www.cancer.gov/cancertopics/factsheet/Risk/acrylamide-in-food>.
- Fogliano, Vincenzo, Simona Maria Monti, Teresa Musella, Giacomino Randazzo, and Alberto Ritieni. Formation of coloured Maillard reaction products in a gluten- glucose model system. Vol. 66. Food Chemistry, 1999. 293-99. 30 Jan. 2014.
- Lersch, Martin. Maximizing Food Flavor by Speeding Up the Maillard Reaction. 2012. Web. 30 Jan. 2014. <http://blog.khymos.org/>.
- Maillard reactions. Food-Info, 2013. 3 Feb. 2014. <http://www.food-info.net/uk/colour/maillard.htm>.